AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
The bohr model of the atom3/10/2024 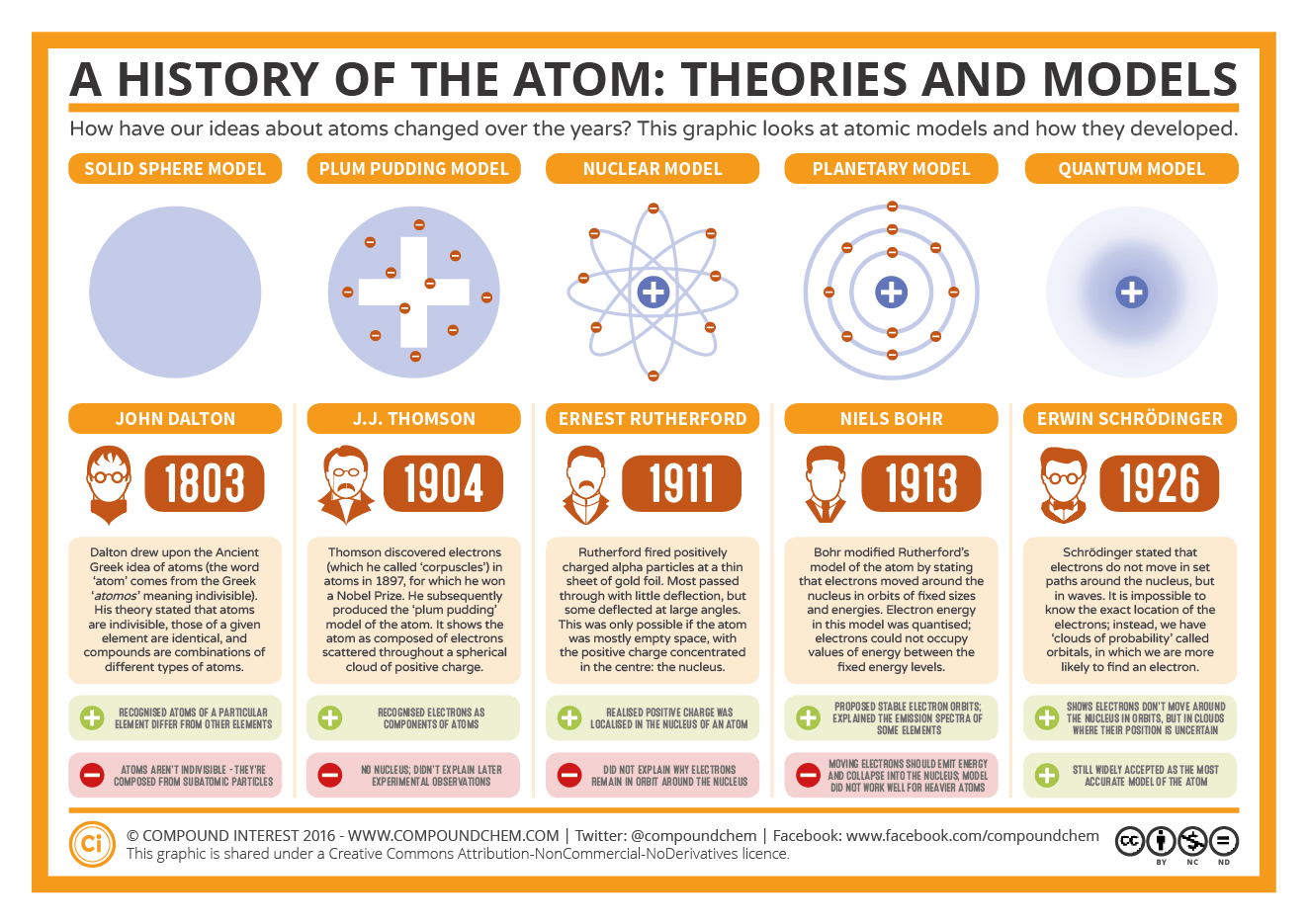
If the atom receives energy from an outside source, it is possible for the electron to move to an orbit with a higher n value and the atom is now in an excited electronic state (or simply an excited state) with a higher energy. The model can be applied to hydrogen or hydrogen-like atoms to explain their line emission spectrum. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. These orbits have quantized energy and angular momentum associated with electron. One of the most important contributions to atomic theory (the field of science that looks at atoms) was the development of quantum theory. Bohr model of an atom states that only those orbits are allowed where angular momentum of electron are integral multiple of n h / 2. Up to 2 electrons orbit in the first energy level. The different orbit distances are called energy levels. In the Bohr model of the atom: Electrons orbit the nucleus at different distances. He used the nuclear model to create his model. The existence of the atomic spectra is support for Bohrs model of the atom. In 1913 the Danish Physicist, Niels Bohr, came up with an improved model of the atom. 
When the electron is in this lowest energy orbit, the atom is said to be in its ground electronic state (or simply ground state). Other models of the atom (ESAAT) Although the most commonly used model of the atom is the Bohr model, scientists are still developing new and improved theories on what the atom looks like. Bohrs model suggests that the atomic spectra of atoms is produced by electrons gaining energy from some source, jumping up to a higher energy level, then immediately dropping back to a lower energy level and emitting the energy difference between the two energy levels. They are: >John Daltons atomic model: Dalton´s Billiard Ball (Solid Sphere) Model >J.J. Thus, the electron in a hydrogen atom usually moves in the n = 1 orbit, the orbit in which it has the lowest energy. There are five basic atomic models which have contributed the structure of the atom itself. One of the fundamental laws of physics is that matter is most stable with the lowest possible energy. Bohr’s model of the hydrogen atom, proposed by Niels Bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
